JAK inhibitors from Pfizer, AbbVie and Lilly hit with dreaded FDA heart safety, cancer warnings | Fierce Pharma
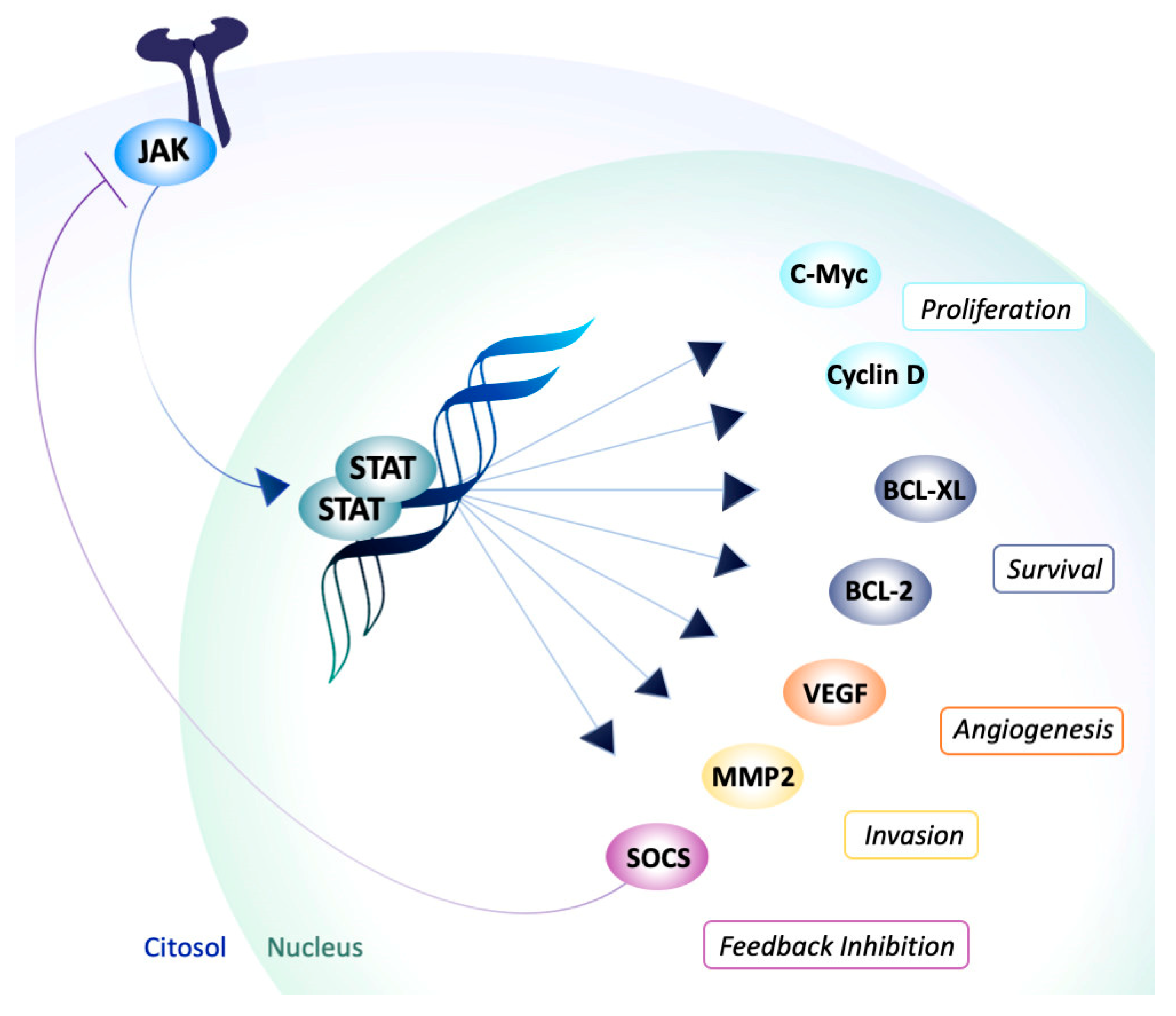
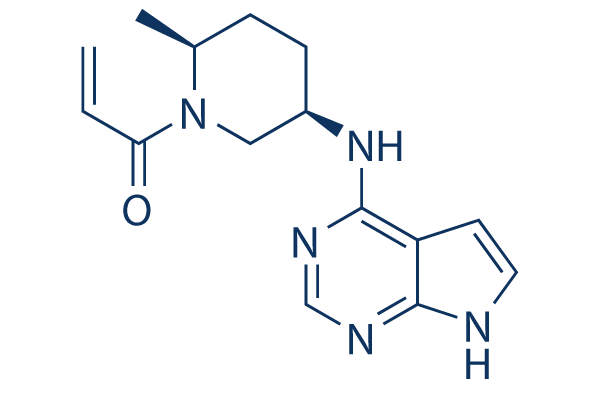
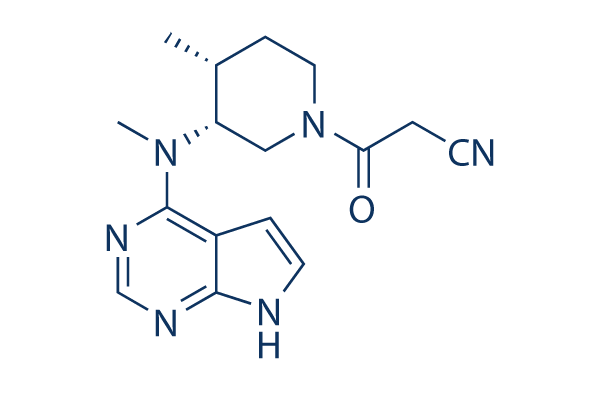
Molecules | Free Full-Text | Inside Perspective of the Synthetic and Computational Toolbox of JAK Inhibitors: Recent Updates

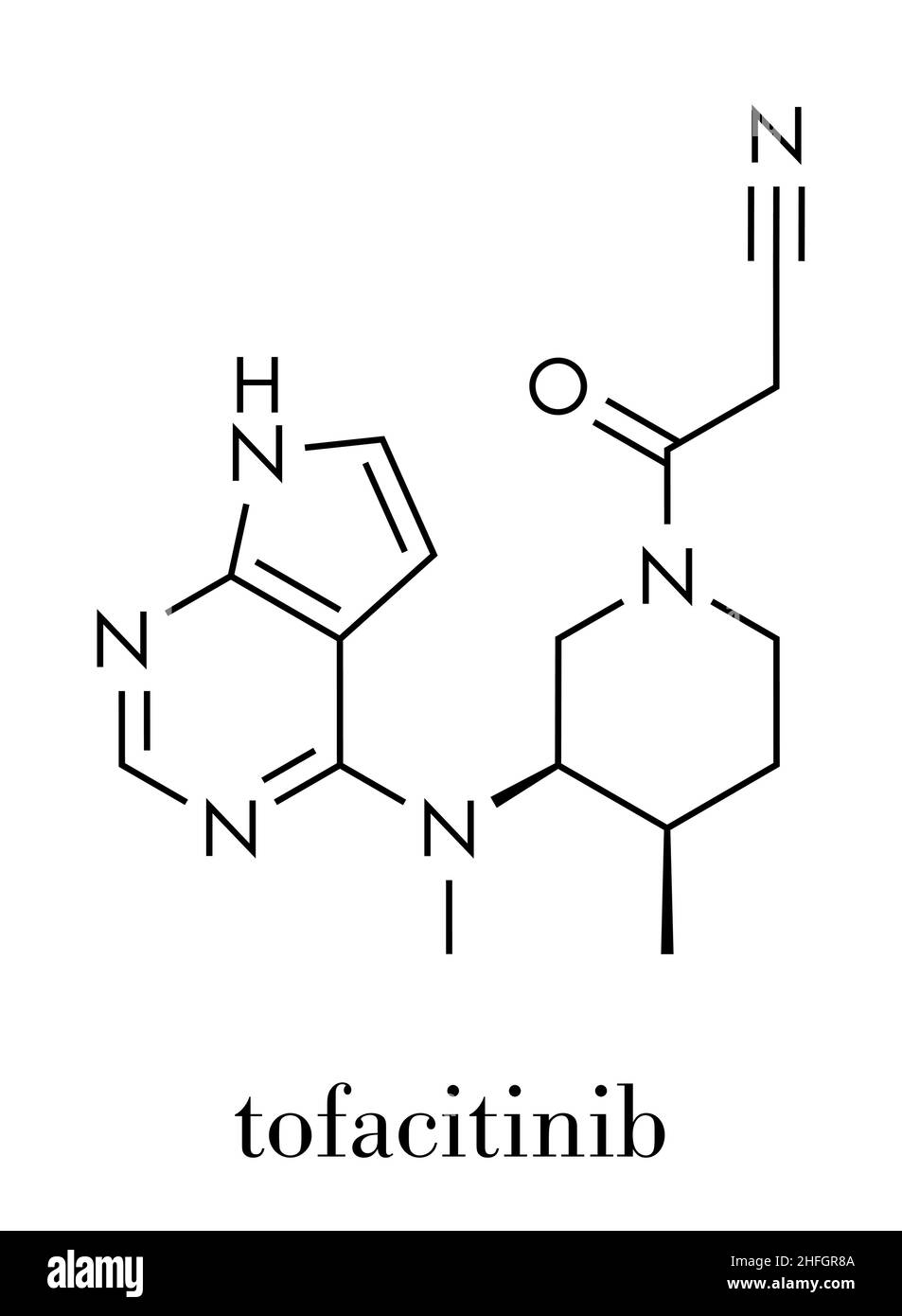
Pfizer Announces FDA Approval of XELJANZ® XR (tofacitinib citrate) Extended-Release Tablets, the First and Only Once-Daily Oral JAK Inhibitor Treatment for Rheumatoid Arthritis | Business Wire

Pharmas with up-and-coming JAK inhibitors face 'shrinking' potential after FDA crackdown | Fierce Biotech
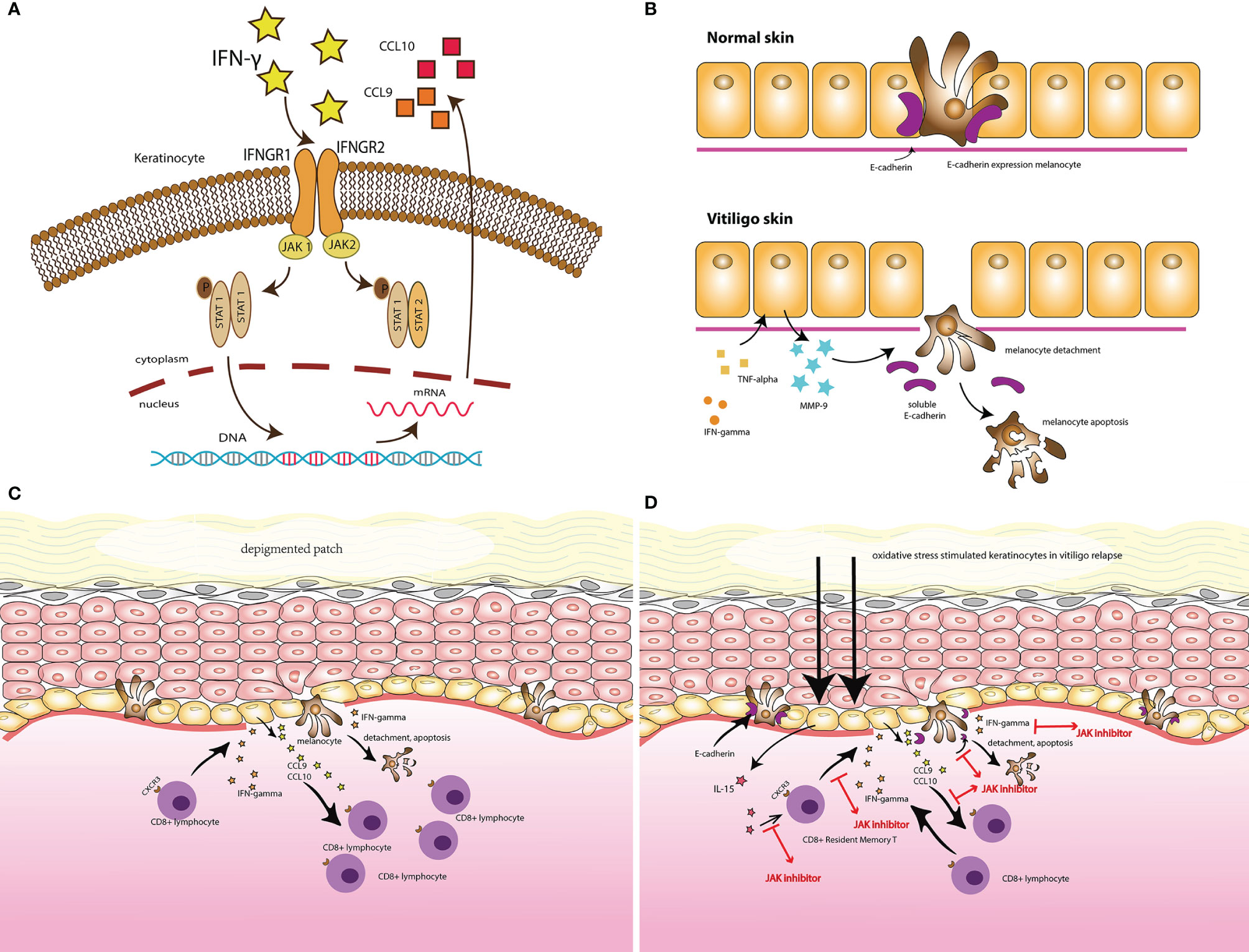
FDA approves JAK inhibitors as 2nd-line systemic therapy in atopic dermatitis < Pharma < Article - KBR

Molecular structures of FDA approved and investigational JAK inhibitors. | Download Scientific Diagram